NEWPORT, CA – Medtrade exhibitor Cure Medical (Booth #950 at Medtrade, scheduled for Oct 23-25 at the Georgia World Congress Center in Atlanta) has unveiled the new 10-inch Pediatric Hydrophilic Cure Catheter® for children who need an easy-to-use, water-lubricated catheter that will not kink when bent.
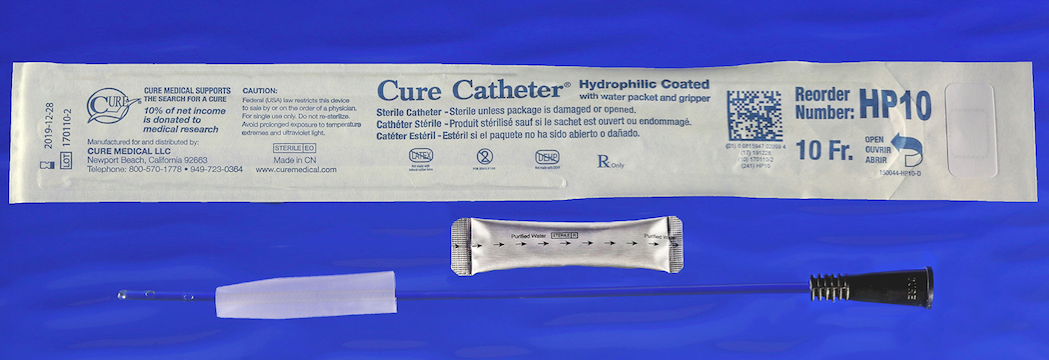
This sterile, single-use hydrophilic catheter features polished eyelets with a straight tip. It is offered in four pediatric lengths for use by boys and girls. The sterile, single use Pediatric Hydrophilic Cure Catheter® offers the following features:
• FOR PEDIATRIC USE
• Sizes: 8-14 French
• 10-inch long, STRAIGHT catheter
• High quality materials — not made with DEHP*, BPA or natural rubber latex
• Smooth polished eyelets for increased comfort
• Purified water packet for easy, mess-free/stain-free instant hydration
• Textured advancer/gripper for clean, controlled use
• The Cure Commitment – Cure Medical donates 10% of net income from every catheter to fund medical research in pursuit of a cure for paralysis and urinary retention.
 The Pediatric Hydropilic Cure Catheter
The Pediatric Hydropilic Cure Catheter
Children often have more severe allergic reactions than adults do, and this is due to their body size. A small amount of allergy-inducing substance often has a higher impact on a 40-lb. child than it will on a 200-lb. man. Finding the right catheter that works for your child can be frustrating, especially when you’re trying to find one that’s safe and easy-to-use.
The Pediatric Hydrophilic Cure Catheter is not made with DEHP, BPA or natural rubber latex – especially for those parents who are mindful about their children’s chemical or allergen exposure. DEHP is included on a published list of chemicals known to cause cancer and reproductive harm according to the state of California. Other manufacturers often use Di(2-ethylhexyl)phthalate (DEHP) as an economical means for making catheters flexible, and this is still legal.
However, if you can avoid these chemicals all together, there’s no reason not to. Cure Medical CEO John Anderson explains, “The use of DEHP in catheters creates a number health risks including patient exposure to hazardous chemicals which leaches from vinyl medical devices.” That’s why Cure Medical products are not made with DEHP.*
Likewise, latex may be in other brands of catheters. Natural rubber latex is a chemical which commonly causes irritation and can cause life-threatening allergic reactions. That’s why Cure Medical catheters are not made with latex. By investing in advanced technology and selecting high quality materials for manufacturing, Cure Medical helps ensure that users experience maximum safety, comfort, and peace of mind.
Free Educational Materials for Parents
In addition to the new Pediatric Hydrophilic Cure Catheter®, Cure Medical is offering a new education program that parents can utilize to teach their children ‘How To Cath’ with fun, instruction tools like:
• Backpacks in orange, red, blue and pink with no logos — for discretion at school or away from home; • Flash cards for boys and girls that also include steps for cathing with or without stomas; and
• Coloring books with crayons, a complimentary toy – and more!
“At Cure Medical, we stand behind our commitments like funding research for a cure by donating 10% of our net Income to research programs worldwide. We also commit to quality care for our customers by including very smooth eyes on every catheter, and we never use obnoxious chemicals like BPA, DEHP [two known carcinogens] or Latex in our products. The CURE NATION is growing every day with delighted customers who are united in pursuit of a Cure, and we invite you to join us too,” Anderson continues.
The Pediatric Hydrophilic Cure Catheter® is available now from Cure Medical and its authorized dealers. To request free samples, contact your local distributor of quality healthcare products or visit http://www.curemedical.com.
* See CA EPA Office of Env. Health Hazard Assessment List of Chemicals Known to the State to Cause Cancer or Reproductive Toxicity, Dec. 8, 2006.



